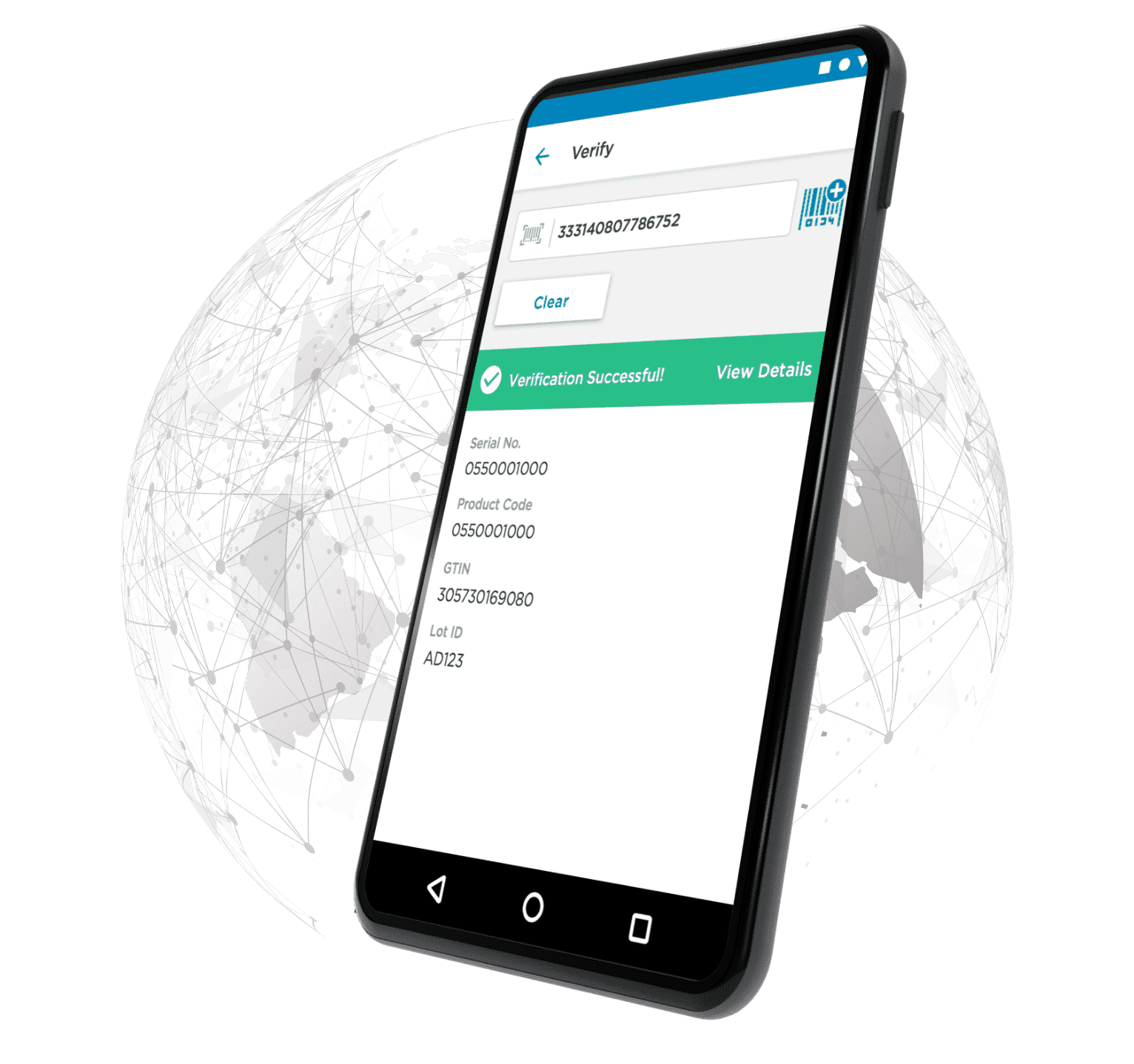
Providing transparency & accountability throughout the supply chain.
- Increased efficiency
- Real-time environmental monitoring
- Smart contracting that produces significant cost savings
- Integration of Blockchain for transaction security
- Integration of Artificial Intelligence for data exploitation
- Interoperable with existing government systems and sensors

Shift the balance of cyber power in your favor with rfxcel’s innovative supply chain traceability software
Acquire
Strengthen networks that distribute goods and services
Prioritize
Critical capabilities, situational awareness, and emergency services
Respond
Know your risks, plan ahead, be prepared
200,000+ successful integrations & connections for customers and their partners
Teams of experts implement & support your solution
Full-service implementation and validation
- Pre-configured and pre-validated solutions simplify your implementation process.
- Receive documented release notes, risk assessments, and detailed test results.
- A fully validated production solution can be implemented in 20–30 days.
Advisory services and support
- We offer 24/7 support to all our customers.
- Our Washington, DC-based team of professionals is on hand to help, advise, and optimize your mission
- A dedicated support person is assigned to address all your inquiries and needs.
- We believe in educating industry players to maximize interoperability.
How rfxcel is leading the digital supply chain for global government
Resilience in Action
By embracing a data-centric security posture with real-time monitoring capabilities, rfxcel enables law enforcement, military, and first responders to proactively handle whatever adversaries do to try to break our supply chain.
Critical Infrastructure
We ensure networks have the tools to communicate, the situational awareness to respond, and the emergency services to recover from a major blow to our critical infrastructure.
Mission Readiness
The complex reality is that modern-day supply chains are vulnerable to attack. With rfxcel, you’ll be ready to respond.
National Security
Supply chain resilience is a matter of national security. With all sectors interconnected, sustainability requires cross-sector inter-dependencies. Are you ready for the next step?
See how Customers are Succeeding with rfxcel

ISO 9001:2015 Certified for Quality Management Systems
rfxcel, the leader in track and trace solutions for global supply chains, achieves ISO 90012015 certification for Quality Management Systems.

rfxcel Will Exhibit at CPhI Middle East & Africa, the Largest Pharmaceutical Event in the Region
rfxcel, the global leader in supply chain track and trace solutions, will exhibit at this year’s CPhI Middle East & Africa (MEA) conference in Abu Dhabi, United Arab Emirates. The company is cultivating business in the region, particularly pertaining to new serialization requirements from the Saudi Food and Drug Authority (SFDA).