
Japan Track & Trace Requirements
Japan’s regulations are included in the Pharmaceuticals and Medical Devices Act (PMDA), passed in November 2014 and revised in December 2019. The PMDA addresses all aspects of medical product registration, including in-country representation, certification processes, licensing, and quality assurance.
About Japan Track and Trace Requirements
Japan is emphasizing “traceability,” not serialization, through the use of GS1 barcodes. It appears the system established in the PMDA requires GS1 DataBar barcodes on primary and secondary packaging, and GS1-128 barcodes on tertiary packaging.
Products that must be barcoded include “specified biological products,” biological products, oral medicines, injectables, topical medications, medical devices, “specified medical materials,” and in vitro diagnostics.
The revised PMDA set two deadlines for barcoding requirements:
- e-labeling (August 2021): GS1 barcodes must be placed on secondary packaging (i.e., “sales packages”); when scanned, they will utilize GS1 Digital Link to connect the user to the PMDA database of all pharmaceuticals and medical devices.
- Barcoding (December 2022): GS1 barcodes must be placed on secondary packaging and include a Global Trade Item Number (GTIN), lot number, and expiry date.
Furthermore, the PMDA is promoting digitization as a means to provide the latest product information to health care workers; however, the government has expressed that uptake may be slow because “some doctors are not familiar with digitization” and “some people need to have information in paper media.”
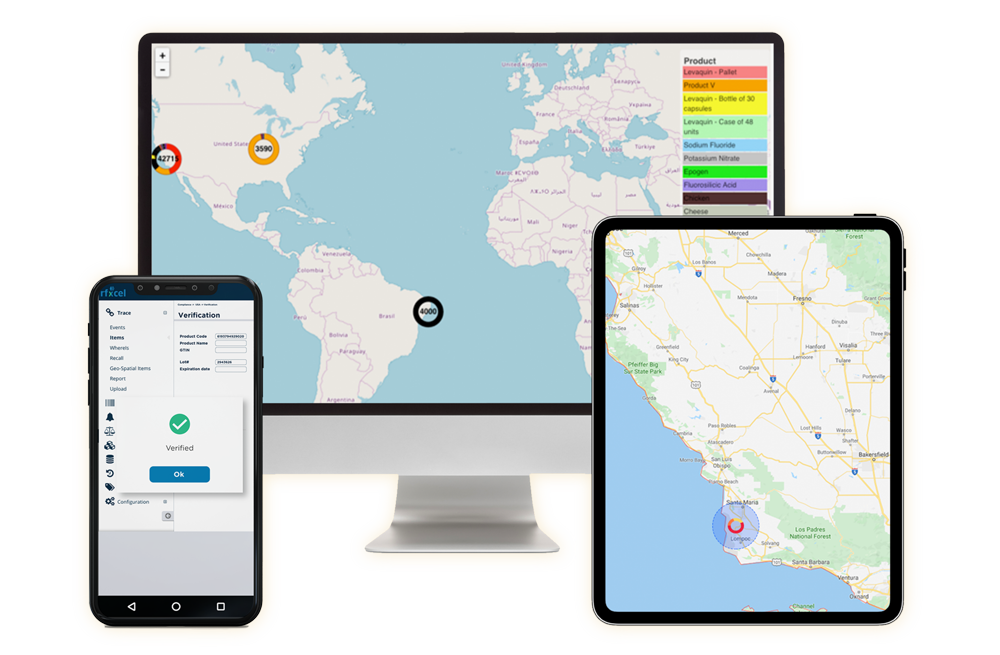
rfxcel Meets Japan’s Serialization & Traceability Requirements
rfxcel Is Your Partner for Japan Compliance
Our team will partner with you to meet Japanese requirements and transform business value with a transparent digital supply chain. With dedicated track and trace solutions from rfxcel, you can safeguard your products and brand reputation by ensuring all items are legitimate, marked, and fully compliant with Japanese law.
rfxcel’s Team: Compliance, Visibility & Support
rfxcel’s team will help you achieve and maintain Japan compliance by bringing end-to-end visibility to your supply chain.
Growth-Centered Solutions
rfxcel will develop a fully validated implementation plan tailored to your needs and designed specifically for Japan. Our systems operate in local languages, local currency, and processes, easing the compliance burden for companies in any industry. Our goal is to increase company value through insightful and precise data by automating and digitizing workflows.
Growth-Driven Support
rfxcel provides 24/7 support to all our customers. We assign a dedicated support team member to our customers so we can quickly and thoroughly resolve any issues that arise.
Secure Your Supply Chain with rfxcel Traceability System
Our award-winning Traceability System is a fully customizable platform that simplifies and accelerates compliance and increases supply chain visibility no matter where you do business. It also integrates seamlessly with your existing systems and enhances scalability by letting you add new features quickly.
Find out more about our Japan compliance solutions.
Your value beyond compliance with rfxcel
- Digitize and centralize your entire supply chain in one elegant, easy-to-use, intuitive platform
- Enhance performance and reduce processing time in multiple languages
- Leverage IoT technology to gain new real-time insight into and control of your supply chain
- Trace your products forward and backward
- Maintain historical information for a batch or individual product with continuous review and supply chain optimization
- Drive growth by lowering costs and risks, eliminating inefficiencies, and tracking all activity anywhere in your supply chain, including among suppliers, distributors, and 3PLs
- Integrate with any trading partner

